- Chinese website
- Sitemap
- Mobile site

- Customer service hotline:400-666-5548

Product Description
Product Name: Medical Isolation shoes cover short
Applicable People:Adult
Standard:YY/T 1633-2019
Material: non-woven fabric material
Place of Origin:Hunan, China
Size:.33*17CM
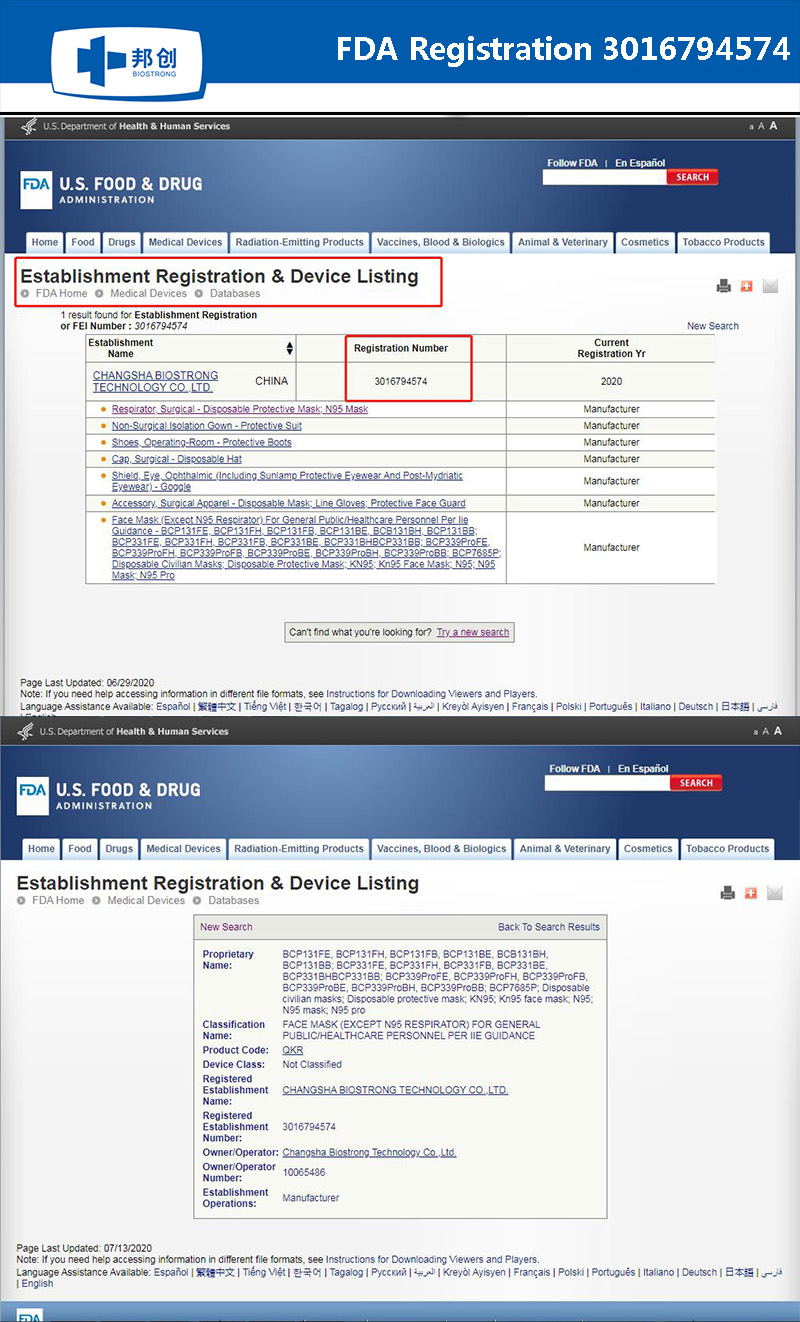
Certificated:FDA Registered
Validity Period:3 Years.
Scope of Application:To provide barrier and protection for clinical medical personnel in medical institutions when they come into contact with the patients with potentially infectious blood, body fluids, and secretions.
Warnings
1.Please check date and package before use. It is strictly prohibited to use when the package is damaged.
2.The product is one-time use only. Repeated use is not allowed. Destroy it after use.
Storage Condition
The package should remain intact, kept away from humid and high temperature, and stored in ventilative place with no corrosive gases.
Direction for Use
1.Unfold the shoe covers before use;
2.Put on the shoe cover;
3.Tuck the socks and trousers cuff into shoe covers(Tie on the band properly if have band )

The manufacturer(MFT) of Pandemic prevention products-Changsha Biotrong Biotechnology Co., Ltd. all rights reserved湘ICP备18002869号-1Technical support:Hnjing
 WeChat
WeChat